Invisible Stuff
I
A PETROLEUM refinery out West had a fire. The plant was engaged in producing gasoline by the cracking process — in which enormous pressures and hot temperatures are harnessed together to convert crude oil into gasoline — when suddenly and mysteriously some part of its high-pressure system gave way. There was a terrific explosion, a volcano of flame, and millions of dollars went up in fire and smoke as the entire works burned to cinders.
So complete was the destruction that not enough metal was left in the cinders to give a clue as to the origin of the accident. But the company had similar refineries in other places equipped with duplicate machinery, and the loss was too serious to risk a repetition. How could a like accident be guarded against over in Kansas and down in Oklahoma? Engineers went through the works in each of these other plants; they searched and tested, and could find nothing wrong. Finally, suspicions centred on a certain steel casting. It looked all right; by every customary physical test it sounded and felt and acted all right; but it occupied a strategic place in the system where the explosion had occurred, and the engineers decided that if there were a hidden weakness in this piece of metal it would cause just such a catastrophe as had wiped out the first plant. So they decided to look at the insides of the casting and see if it was sound all the way through.
A few years ago there was just one accepted way of inspecting the interior of a piece of metal. You could break it or saw it into sections, and thus open its inner structure; but of course after you had seen it, and perhaps proved that it was sound, you would have destroyed the object itself. Such testing is like striking a match to see if it is good.
The petroleum engineers did not like the idea of sawing up this casting; it was too important, too costly, its replacement would mean expensive delay. They appealed to a government institution, the Watertown Arsenal in Massachusetts, which had been using X-rays to inspect steel castings for army gun carriages. The X-ray expert there, Dr. H. H. Lester, told them to send on the casting; and a few days later he was able to show radiographs (X-ray photographs) of it.
The pictures scared the oil men. What did this black streak near the centre mean?
‘It means an interior crack,’ answered the physicist.
A crack in a section that must endure tons of pressure! It was what they suspected, of course, but hard to believe, just the same.
They sawed the casting in two, and there was the crack, exactly as indicated by the radiograph. The X-ray inspection was completely vindicated. You may be sure that no more castings were installed by this company until they had passed the X-ray test.
To most readers it will come as a surprise, perhaps, to learn that this delicate, highly penetrating radiation, which is used by the surgeon in setting a broken bone and by the dentist in exploring a troublesome tooth, has now rolled up its sleeves and gone into the factory and taken its place as one of the tools of production. There are certain industrial plants where X-rays are being used regularly to inspect three and one-half to four inches of steel, and in research laboratories even greater penetration has been attained experimentally. The idea is not new, but its applications are, and the last year has developed many new applications. Altogether, more than twenty-five industrial plants in the United States are now using X-rays in the inspection and testing of materials and products — and many more are putting to work in their manufacturing processes the new knowledge of metals and other materials recently discovered in the X-ray laboratories.
II
Industrial possibilities of X-rays were early suggested by the discoverer himself. In his very first paper on the subject, published in 1895 just a few weeks after he had first beheld the strange luminescence of barium salts when penetrated by the unknown radiation, Dr. W. K. Röntgen reported various objects that he had radiographed. Among them was ‘a piece of metal whose lack of homogeneity becomes noticeable by means of X-rays.’ It is precisely this that Dr. Lester and the other industrial radiologists are checking up to-day by means of the rays. For any lack of homogeneity in a part that has to endure stresses and strains is a sign of weakness. In our machine age of high speed and high pressure and high temperature, such signs had best be discovered before, rather than after, using.
The World War demonstrated the industrial value of X-rays as a detective device. Down at Galveston, for instance, cotton shippers during the early war months made X-ray photographs of bales destined for Germany, and thus were able to satisfy the clearance officers that there was no copper or other metallic contraband concealed within their cotton. Later, when the United States got into the war, X-rays began to be used at our munitions factories to inspect the finished bombs, grenades, detonators, and other complicated devices, to make sure that their parts were complete and accurately assembled. Then, after the munitions were packed and delivered to the docks, the sealed cases were X-rayed to check against shortage, thievery, and sabotage.
Meanwhile, abroad, this new inspection had developed earlier and on an even more comprehensive scale. Much of the modern technique of industrial radiology was worked out then by the British physicists, Pullin, Kaye, Knox, and their associates in the British war service.
One day a new airplane, one of several recently received from the same manufacturer, snapped an axle of its undercarriage in landing. The ruined part showed that the break had occurred at a place where a hole had been drilled through the axle and then plugged up and cleaned off to conceal the flaw. The question immediately arose, How many other planes had been similarly treated? All of the remaining axles were radiographed, and several were found to be faked in this same way — though the most expert visual examination failed to find the plugged holes.
Not only aircraft metal, but also the vital wooden parts were subjected to the X-ray test. Thus there was the case of a split spar in which the workman had skillfully glued a strip of shaving over the crack. Sandpapering completely concealed the defect, and this dangerous section would undoubtedly have passed inspection but for the X-rays. Under their imperturbable invisible glow the line of that crack and the shadow of its glued covering loomed up in the radiograph as unmistakable as the hole in the doughnut.
Hidden knots, resin pockets, and other imperfections of the wood, peculiarities in the structure of the fibres, the differentiation between heartwood and sapwood, between summer and spring growths, the presence of wormholes and even of the entombed bodies of dead larvæ — all these hidden differences and defects were clearly revealed. Indeed, the air ministries of the Allies found that one of their most facile tools in providing wings for the fighting forces in France was this mysterious glass bulb which had been hastily requisitioned from the hospital and the research laboratory to provide its unfoolable, unbribable inspection.
The modern aircraft industries would be seriously handicapped if deprived of X-rays. Aviators are more and more demanding X-ray inspection of their machines. The engine of the biplane Bremen, which made that spectacular flight from Germany to North America, was radiographed in all its vital parts before it ventured the Atlantic west wind. Several aircraft manufacturers will accept no castings that fail to pass the radiographic test.
On the sea, as in the air, the X-ray is becoming an accepted insurance device. When the cup defender Enterprise was under construction, all the steel forgings that went into her trim and elegant hull were scrutinized by means of the rays, and had to pass that. inspection before they were accepted.
A boiler factory in the Middle West installed in 1930 an X-ray apparatus which is mounted on wheels and travels along a rail. In this way it is able to radiograph a strip of welding many feet long, and to determine the soundness of a seam in a huge boiler or a long tank car.
A cheese factory is using X-ray inspection to make sure of the size and position of the holes in its product — apparently holes are an important essential of proper cheeses! Rubber factories are using the same means to prove the symmetry of the cores of golf balls, to determine the quality of rubber tires, and to inspect the imbedding of metal washers in rubber heels. In another factory, scrap rubber is sorted through X-rays to detect any metal filings or other foreign waste before it is put back into the pot to be remelted and reprocessed. The vacuum tube with its hidden wiring and delicate construction, the telephone cable with its thousands of insulated strands, the lump of coal with its varying content of slate and ash — all these are inspected and passed or rejected on the ‘ say-so ’ of X-rays.
Even art has its industrial demands on this invisible light. At the world conference of art experts, held at Rome in 1930 under the auspices of the League of Nations, many scientific tests to fix the genuineness of works of art were reported. Dr. Paul Ganz, of the University of Basel, told of discovering through X-rays a precious Holbein under a painting of no particular value. The old masters used mineral pigments, which are more opaque than our modern vegetable pigments; therefore a canvas on which a later work has been painted over an old one will almost invariably reveal its hidden original when photographed by X-rays.
On another occasion, Dr. Ganz discovered that a portrait of Sir William Butt, the younger, was in reality a Holbein on which a later artist had painted whiskers and wrinkles to represent the proud knight as an elderly man. But X-rays know nothing of vanity or of proud purses. They betray imitations as well as reveal the hidden treasure. Mr. Alan Burroughs, who is in charge of the extensive X-ray work at the Fogg Art Museum of Harvard University, tells me that spurious art claims have been punctured and fraudulent sales prevented through the telltale evidence of X-rays.
This American museum has made itself a leader in the application of X-rays to art studies. It has not only radiographed practically all of the paintings in its own valuable collection in Cambridge, but has extended these studies to many other collections on both sides of the Atlantic. As a result, it has assembled eighteen hundred radiographs of art works, including many of the most esteemed paintings in the Louvre, the Kaiser Friedrich Museum, the National Gallery in London, the Royal Museums of Brussels and Antwerp, and the Museum of Bruges, Coöperating in this country have been the Metropolitan Museum in New York, the Cleveland Museum, the Art Institute of Chicago, and the Boston Art Museum. Classification of the information thus obtained by X-rays has already begun, in the hope of settling eventually some of the vexed questions which confront connoisseurs almost everywhere as to the authenticity of disputed paintings.
Another interesting application of X-rays has recently been reported from the Fogg Museum. In this case it was desired to restore some rare bronzes brought by a collector from Iraq. The objects were just as they had been dug out of the ruins, heavily encrusted with sand, clay, and corrosion; and the success of their restoration by the electrolytic process depended on whether there remained any solid metal core. By means of X-rays these ancient relics were photographed, the extent of their remaining metal was shown, and thus the technicians were guided in what has proved to be a very satisfactory restoration.
Police have used X-rays to search jewel thieves. The same means have been employed in the South African diamond fields to detect diamond swallowers among the miners. There is an instance reported from England in which an elephant was radiographed in the search for a valuable jeweled ring which he had swallowed. A French expert secured radiographs of thumb prints by rubbing the thumb of the subject with red lead before photographing. The resulting shadow picture shows not only the surface lines, but also the shape of the thumb bone, thus providing an added detail of identification.
It is now possible to make such sharp radiographs of the skull that one authority suggests their use to supplement thumb prints as a means of identification of persons. Recently biologists were baffled by a disease affecting carp in the Illinois River. Dissection methods were not successful in diagnosing the obscure ailment; but with the aid of X-ray pictures of the afflicted fish the scientists were able to identify the trouble positively. Applications of X-rays to human diagnosis are as numerous as are their applications to industrial inspection.
Nor is that all. Just as the surgeon, by his radiographs of the broken bone and its progressive changes under his treatment, is enabled to improve his surgical methods, so the industrialist is finding in the X-rays a guide to new and better manufacturing technique. Perhaps the most important contribution of the X-ray laboratory which the Government established at the Watertown Arsenal in 1922 is the improvement in metal-working processes which has resulted from this pioneering in a new form of inspection. For the radiograph is used here, not only to discover defective castings, but also to test new methods of making castings, and thus to establish improved processes.
So great had been the improvement of castings under this system that when the Arsenal in 1928 decided to try the fabrication of gun carriages by means of electric welding the engineers immediately resorted to X-rays to inspect the welds. By varying their methods under this all-seeing eye of the sensitive film, they were able to develop a new welding technique.
It was found, for instance, that a certain energy of the electric arc gave the best results. When the current was greater than this, porosity in the welds began to show in the radiographs; when the current was less, the X-rays revealed a lack of fusion and adhesion between the welded surfaces. Similarly, by X-raying each trial, they learned that for one particular job six inches a minute was the right rate of feed. So, too, they checked up on the right size of welding rod — and, in fact, on every detail of the process. A good weld — which used to be described (like Bernard Shaw’s definition of a good play) as a weld that was made by a good welder — now was reduced to established standards. Naturally, the percentage of work that needs to be rewelded has grown less and less. And now the art of welding may be said to have arrived at a scientific basis — thanks to the shadow pictures which Professor Röntgen thirtyfive years ago showed us how to make.
III
Meanwhile, the laboratory explorers have found that the X-ray can do more than provide shadow pictures. New powers have been discovered — capabilities which Röntgen could hardly have suspected. For while the immortal professor inferred that his rays were something different from the stream of electric particles which generated them, he did not know what they were. We cannot say that we know fully what they are to-day; but at least we are sure that they are of the same nature as light rays, being of a much shorter wave length and obeying the laws of electromagnetic radiation which Clerk Maxwell formulated theoretically fifty-odd years ago. In spite of Maxw ell’s prediction of the existence of radiation of short wave length, it was not until 1913 thatX-rays were identified as belonging to this category.
Since then, however, their application has fairly galloped. The modern physicist is using X-rays to explore the molecular and atomic structure of things — to identify, measure, and chart values far beyond the reach of the most powerful microscope. By means of X-rays he has discovered a new chemical clement, hafnium. By means of X-rays he has explored the inwardness of solids — of which we know far less than we know of liquids and gases — and has discovered the fundamental crystalline structure of all solid matter. By means of X-rays he has found that the muscular tissue of human flesh is made of minute crystals, that wood and cotton and silk and all natural fibres are crystalline, that the varying properties of metals are an expression of varying arrangements of their crystals.
Nor have these research results stayed in the laboratory. Industry has promptly taken them up and applied them in its manufacturing processes. Thus, at the University of Illinois, Dr. George L. Clark has been studying the structure of fibres through the patterns which they reveal on a photographic film by their diffraction of X-rays. Dr. Clark found that silk gives a pattern different from that diffracted by rayon. From these studies he was able to draw a picture of the crystalline structure of the two fibres, indicating the size and arrangement and characteristics of each. One of the manufacturing companies took these results, and by using X-ray diffraction to test each step of its process it has succeeded in spinning a new rayon which closely approximates silk in the size, strength, tenacity, and elasticity of its fibre.
At the X-ray laboratory of the Massachusetts Institute of Technology, Professor John T. Norton showed me photographs made by metals through this same phenomenon of X-ray diffraction. Each metal had written its autograph on the film in a symmetrical pattern of concentric rings, each ring and segment telling the physicist something of the arrangement of atoms into crystals. These structures are infinitesimally small. Thus the iron crystal, which is a cube with an atom of iron at each of its eight corners and a single atom inside the cube at its exact centre, measures less than three two-hundred-millionths of an inch across its face. The eye can never hope to see these minutiae; light rays, by which we see, are too large and clumsy for such microcosms. But by the deflected X-rays, bent as they strike the planes and lattices of the atomic crystals, the physicist is enabled to visualize these structures so exactly that one may fairly say he ‘sees’ them.
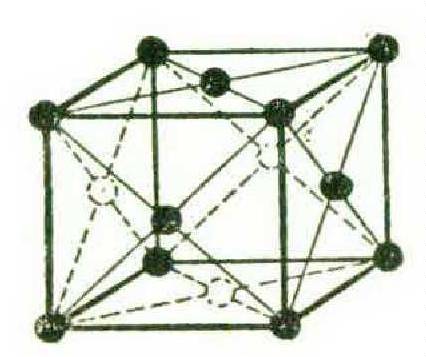
X-rays reveal that most common metals are of three crystalline forms. The soft ductile metals — copper, silver, gold, aluminum, and the like — are built up in cubes with an atom in each corner and an atom centred in each face, like this: —
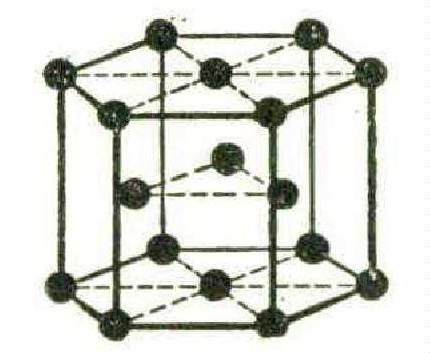
Zinc, magnesium, beryllium — less ductile and more brittle than the facecentred metals — have their atoms in the form of a close-packed hexagon, in this wise: —
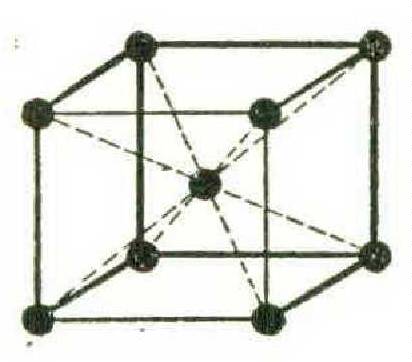
Finally, there are the body-centred crystals, in which iron, tungsten, molybdenum, chromium, and the other hard metals arrange their units — cubes with the corner atoms tightly held by the inner centred atom: —
Scientists long played with theories as to the fine structure of metals, but it is only recently that X-rays revealed unmistakably the simplicity of these three building blocks. Copper thus has the same form of crystal as gold, silver, and aluminum, but differs in size. In the aluminum crystal the atoms are more than 10 per cent farther apart than those of the copper crystal.
X-rays show what happens to a metal when it is subjected to different conditions. Just as a man’s handwriting may reflect his sobriety or fatigue or excitement, so the X-ray diffraction pattern reveals the effect on the crystal of rolling, annealing, heat-treating, and alloying.
When copper is fused with zinc, the diffraction pattern shows the picture of the alloying process — and the expert can fairly see the tiny hexagons of zinc edging into the cubes of copper to form brass. Similarly, the X-ray shows why tungsten carbide is harder than steel, why cold-rolling a plate makes it hard, why heat-treating a metal relieves its strain.
Several years ago Dr. Ancel St. John, studying carbon by X-ray diffraction methods, discovered a new form of the element — a crystalline carbon similar to graphite in its electrical conductivity, but having a higher resistance and being an abrasive rather than a lubricant. One of the manufacturing companies has found this discovery useful in the manufacture of an electric product. By the same method of diffraction analysis, this physicist was able recently to settle a disputed point in a patent suit. The question was whether in a certain appliance the element thorium was present in its metallic state or as thorium oxide. The diffraction pattern of the compound differs from that of the element, and by the X-rays Dr. St. John established positively that the element was present as a metal.
All of these applications of diffraction analysis are derived from the brilliant discovery by Laue, a German experimenter, in 1913. Prior to this, investigators in many lands had tried prisms and finely ruled gratings in the attempt to break X-rays up into a pattern of wave lengths similar to the rainbow pattern which light gives when it is diffracted. But the X-rays passed in straight lines through all prisms and gratings, or, if the material was dense enough, the rays were stopped — and no diffraction could be noted. It occurred to Laue that Nature itself had provided in the fine lattices of atomic crystals a grated surface of a fineness sufficient to deflect the minute wave lengths of X-rays. He tried the experiment — and his success is history. (One should add, to bring history up to date, that recent experimenters with optical gratings have succeeded in diffracting X-rays. Notable among these doers of the impossible is Dr. Arthur H. Compton, of the University of Chicago, whose X-ray discoveries brought him the Nobel prize in 1927.)
It was found that X-rays vary in wave length from about one hundred-millionth to about one ten-billionth of an inch. They were emitted in Röntgen’s original experiments from the glass surface of the bulb, generated by the impact of the speeding particles of the electric current as they bombarded the atoms of the glass.
The next step was to place a metal target in the path of the speeding particles; and it was later discovered by the English experimenter Moseley that the X-rays are characteristic of the target material. Thus a copper target gives a different wave length from that of a platinum target, and so for each material. Hence a new means of analysis was placed in the hands of the scientist. If he was given an unknown substance to identify or analyze, it was necessary only to place a sample of the substance on the X-ray target and turn on the current. If the substance was a known element or compound, its radiation would betray it just as unerringly as his thumb print betrays the human suspect. And just as helium was discovered in the sun by spectrum analysis of sunlight, so the rare element hafnium was discovered on the earth by spectrum analysis of the X-rays generated by the unknown substance.
IV
The effect of X-rays on life is less known and not so well formulated as is their effect on such inanimates as the crystals of steel and silk and rayon. For many years hospitals and medical research institutes have been using X-rays to treat cancerous and other malignant growths, much as they use radium. It has not been possible as yet to generate X-rays of the same power as the gamma rays of radium, but X-rays may be produced at will and at relatively less expense than radium radiation. The dangers involved in exposure to these intense transfers of energy should be obvious. Only recently, in 1930, another name was added to the roster of martyrs to X-ray research, when Dr. Edward Mulvaney, medical inspector for the Jersey City Health Department, died as a result of X-ray burns. It should be mentioned, however, that his original burns were contracted several years ago, before the nature and danger of this radiation were realized. In modern practice, the X-ray instrument is screened in lead, and some laboratories devoted to X-ray work present the appearance of heavily armored vaults. Eight tons of lead cover the inner walls and ceiling of the industrial X-ray laboratory at the Massachusetts Institute of Technology.
While medical men have been experimenting with the therapeutic uses of X-rays, the research biologists have been trying them as a key to unlock the mystery of evolution. Why should a species change? If characteristics are inherited, why should there be any break? Why should not sons and daughters be exactly like their fathers and mothers and grandfathers and grandmothers, on down through the generations ?
In the fall of 1926, Professor Hermann J. Muller, of the University of Texas, began a series of experiments with such questions in mind. He placed fruit flies (Drosophila) in gelatin capsules and exposed them to X-radiation. Then he allowed the flies to breed, and observed the resulting progeny.
The children born of the X-rayed fruit flies were, to quote Dr. Muller, ‘startling and unequivocal. All types of mutations, large and small, ugly and beautiful, burst upon the gaze. Flies with bulging eyes or with flat or dented eyes; flies with white, purple, yellow, or brown eyes; flies with curly hair, with ruffled hair, with parted hair, with fine and coarse hair, and bald flies; flies with swollen antenna?, or extra antennæ, or legs in place of antennae; flies with broad wings, with narrow wings, with upturned wings, with downturned wings, with outstretched wings, with truncated wings, with split wings, with spotted wings, with bloated wings, and with practically no wings at all. Big flies and little ones, dark ones and light ones, active and sluggish ones, fertile and sterile ones, long-lived and short-lived ones. Flies that preferred to stay on the ground, flies that did not care about the light, flies with a mixture of sex characteristics, flies that were especially sensitive to warm weather. They were a motley throng. What had been done? The roots of life — the genes — had indeed been struck, and had yielded.’
It sounds like a ghastly orgy of mutilation — but scientists ask whether it may not be a demonstration of the method of evolution. May not the changes which we see in nature be brought about by the radiation which is continually bombarding all living things on this planet — radiation from star stuff and from radioactive elements in the earth? Since this question was first raised, Dr. Muller and Dr. L. M. Mott-Smith, of the Rice Institute, have collaborated in a study of this possibility. They find that the cosmic and other radiation carries energy sufficient to cause only a fraction of the mutations which occur. If their findings hold, it would seem that radiation is only one of the factors that change the genes. The direct causes may be chemical, within the very molecules or atoms of the genes themselves, with the rays serving only as an accelerating agent. If so, certainly they are the most powerful accelerator of the kind known. Dr. Muller finds that normally, in nature, changes occur in about five individuals out of a thousand, whereas the most powerful X-ray treatment produced changes in about seven hundred out of a thousand.
Meanwhile, others have tried the rays on other forms of life. Dr. Stadler, at the University of Missouri, observed marked changes in barley and corn grown from X-rayed seed. At the University of California, Dr. T. H. Goodspeed X-rayed tobacco seeds; they sprouted into a curious medley of tobacco plants — dwarfs, giants, some stubby and thick-leaved, others slender and tall, some with curiously inverted leaves.
’Whether the biologists will be able to control these mutations — to produce finer tobacco, better barley and corn, more capable fruit flies, fatter pigs, and superior men — is something yet to be proved. A plant physiologist in New Jersey claims that he improved his yield of potatoes by X-raying the potato seeds. Plants grown from these treated seeds flowered and fruited one to three weeks earlier than those grown from untreated seed, and their tubers were larger and more plentiful.
At all events, the X-ray tube has gone into biology. The gardener and the stock breeder of the future may find it as invaluable in solving their problems as the foundryman and the textile manufacturer are finding it to-day in solving theirs.
V
But the industrialists are not satisfied. To quote one: ‘The persistent tendency in industry, buildings, bridges, machines, is toward greater size, greater power, greater output; these developments demand steel that can be depended on for super-performance.’
For example, there is now under construction a turbine engine which will take steam at a pressure of 1200 pounds to the square inch and discharge it into a vacuum. The turbine is designed to drive a dynamo which will generate 250,000 kilowatts of energy. This is enormous power — the total connected load of the city of Boston in 1930 was only around 200,000 kilowatts. The cost of interrupting so much production even for a few minutes becomes prohibitive, therefore the 250,000-kilowatt generator cannot be shut down to repair or replace a broken forging or casting — it must be of such steel that it can be counted on to be accident-proof.
Continued increases in the size of machines and structures mean eventual increases in thickness of the steel parts. Already there is a demand for steel castings of six to ten inches cross-section, and X-rays as generated in commercial practice to-day are not able to radiograph satisfactorily through such densities. About four inches is the upper limit for the most powerful tubes available to industry.
To overcome this limitation and inspect sections of the greater thickness required by the United States Navy, scientists at the Naval Research Laboratory recently tried the gamma rays of radium as a substitute for X-rays. Since gamma rays have a wave length from one tenth to one twentieth that of X-rays, they are more penetrating. The naval researchers succeeded in photographing through twelve inches of steel, and they made exceptionally clear pictures through ten inches.
‘I don’t see any necessity for going much beyond this,’ said Dr. R. F. Mehl, who conducted this notable industrial research, aided by Dr. C. S. Barrett of his staff and Dr. G. E. Doan of Lehigh University. ‘ But if industry requires it, I am confident that we can radiograph through even thicker sections. We may increase the penetration either by increasing the time of exposure or by increasing the amount of radium (or radium emanation) used.’
This novel industrial application of radium — which was first announced in the fall of 1930 — has been on trial at the Laboratory near Washington since early in 1929. Dr. Mehl and his associates made many comparative tests of gamma radiation and X-rays, and claim for the former these advantages: (a) its simplicity, the process requiring no high-voltage wires or complicated apparatus, only a capsule of radium or emanation; (b) the possibility of taking many radiographs simultaneously, since the radium radiates continually and equally in all directions; (c) the portability of the radium set-up; (d) its power to radiograph irregular sections of steel; (e) its higher penetration.
It was found that a modern X-ray tube operating at a given current would require ten minutes to photograph through three inches of iron; for the same tube under the same operating conditions to photograph through six inches would require an exposure five thousand times as long — or more than thirty-four days of continuous radiation. By contrast, to increase the thickness of a gamma-ray inspection from three to six inches it was necessary to increase the period of exposure only ten times — a matter of hours to compare with the more than a month required by the X-rays.
Thus radium is beginning to follow its elder brother, the X-ray tube, out of the hospital and laboratory into the foundry, the factory, the inspecting room. Already the Navy has put radium emanation to work in a programme of inspection. And I am informed that one of the large commercial foundries in the United States has adopted gamma-ray inspection for its heavy steel castings. The report of the naval research attracted wide attention in the steel industry here and abroad, and commercial applications of its results are sure to multiply.
VI
And so one may say with reason that the machine is harnessing to its uses even the invisible stuff of radiation. The word ‘stuff’ is not loosely chosen. One of the paradoxes of modern science is the discovery that these transfers of energy which we call rays are not only waves in space, but are also particles, tiny bundles of energy which behave in some experiments like bullets. When these bullets pass through the spaces between atoms and between the electrons of an atom to a photographic film, they affect the chemical coating of the film much as light affects it in our pocket cameras. But when one of the bullets happens to strike an atom or one of its electrons, the bullet may be stopped, absorbed, converted into new radiation. Obviously, the thicker and denser the material penetrated, the greater the number of bullets that will happen to hit and be absorbed. And wherever there are holes, cracks, porosity, or inclusions of less density, the more bullets will get through to the film, and so the greater will be its exposure. It is in this way that the invisible stuff helps us to see the hidden interior of the more ponderable visible stuff.
The same invisible stuff, hitting through and against the lattices of atomic crystals, shows their structure and size — betrays their unseen make-up.
And the same invisible stuff, bombarding a living cell with its bullets, knocks out parts of the cell’s atoms, and so changes the very nature and characteristics of the life unit itself.
All these powers of radiation have been discovered within the last three and a half decades, most of them within the last two. Perhaps another few years will bring confirmation of that bold hypothesis which Dr. R. A. Millikan has suggested: that the invisible stuff of radiation is the creative force in nature by which matter is made and the elements forged into their characteristic forms. A deus ex machina — or, more accurately, a deus in machina — continually rewinding Time’s clock.